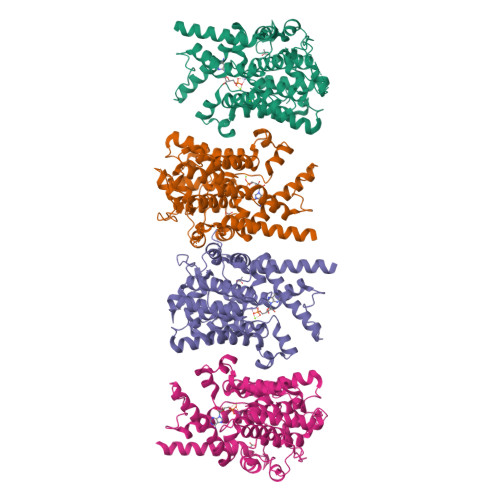
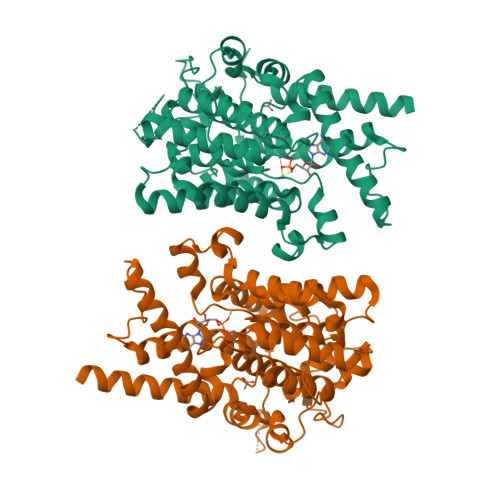
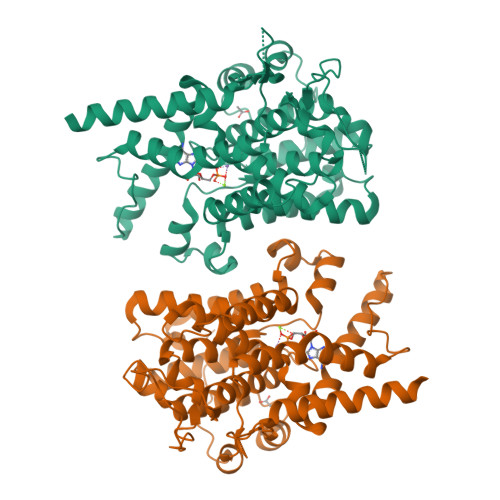
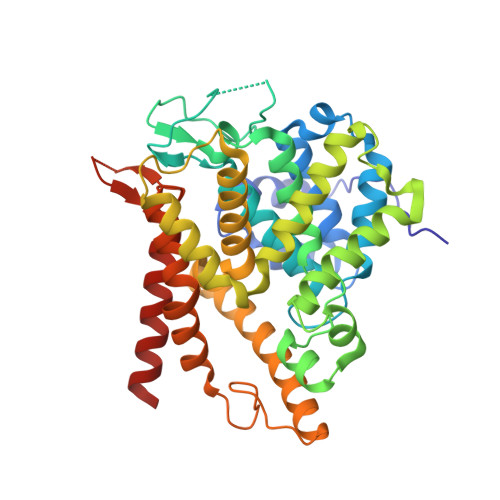
Structure of PDE3A-SLFN12 complex reveals requirements for activation of SLFN12 RNase.
Garvie, C.W., Wu, X., Papanastasiou, M., Lee, S., Fuller, J., Schnitzler, G.R., Horner, S.W., Baker, A., Zhang, T., Mullahoo, J.P., Westlake, L., Hoyt, S.H., Toetzl, M., Ranaghan, M.J., de Waal, L., McGaunn, J., Kaplan, B., Piccioni, F., Yang, X., Lange, M., Tersteegen, A., Raymond, D., Lewis, T.A., Carr, S.A., Cherniack, A.D., Lemke, C.T., Meyerson, M., Greulich, H.(2021) Nat Commun 12: 4375-4375
- PubMed: 34272366
- DOI: https://doi.org/10.1038/s41467-021-24495-w
- Primary Citation of Related Structures:
7KWE, 7L27, 7L28, 7L29, 7LRC, 7LRD, 7LRE - PubMed Abstract:
DNMDP and related compounds, or velcrins, induce complex formation between the phosphodiesterase PDE3A and the SLFN12 protein, leading to a cytotoxic response in cancer cells that express elevated levels of both proteins. The mechanisms by which velcrins induce complex formation, and how the PDE3A-SLFN12 complex causes cancer cell death, are not fully understood. Here, we show that PDE3A and SLFN12 form a heterotetramer stabilized by binding of DNMDP. Interactions between the C-terminal alpha helix of SLFN12 and residues near the active site of PDE3A are required for complex formation, and are further stabilized by interactions between SLFN12 and DNMDP. Moreover, we demonstrate that SLFN12 is an RNase, that PDE3A binding increases SLFN12 RNase activity, and that SLFN12 RNase activity is required for DNMDP response. This new mechanistic understanding will facilitate development of velcrin compounds into new cancer therapies.
Organizational Affiliation:
Center for the Development of Therapeutics, Broad Institute of MIT and Harvard, Cambridge, MA, USA.